The rapid popularization of wearable electronics, soft robots and implanted medical devices has stimulated extensive research in flexible batteries, which are bendable, foldable, knittable, wearable, and/or stretchable … Different from the conventional batteries that utilize rigid and bulky electrodes, current collectors, metal anodes, liquid electrolytes, and packages, flexible batteries require the flexibility of each component to accommodate diverse shapes or sizes.
Metal–air batteries are considered a suitable option for flexible batteries. Oxygen, O2, in the air reacts with a metal electrode in all cells. Sodium, Na, magnesium, Mg, aluminium, Al, and zinc, Zn, are being investigated using polymer and gel electrolytes. A diagram of one of the cells in a flexible zinc–air battery is shown below
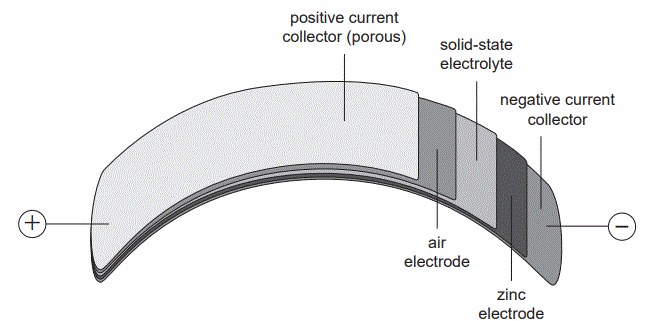
1.Which one of the following shows the migration that occurs through the polymer/gel electrolyte when the flexible cell is producing electrical energy?
A. Electrons flow from the anode to the cathode.
B. Metal ions flow from the anode to the cathode.
C. Electrons flow from the cathode to the anode.
D. Metal ions flow from the cathode to the anode.
Solution
2. Which one of the following cells being investigated for use in flexible applications has the highest potential difference?
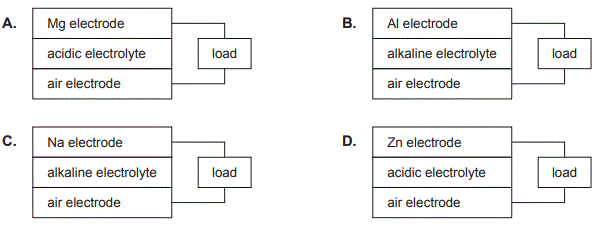
Solution
3. Unwanted side reactions may occur in metal–air cells when moisture is present. This is less likely in
A. Al–air cells.
B. Mg–air cells.
C. Na–air cells.
D. Zn–air cells.
Solution
4. In a nickel–cadmium cell, the following reaction occurs during discharge.
2NiO(OH) + Cd + 2H2O → 2Ni(OH)2 + Cd(OH)2
Which one of the following represents the half-equation for reduction during recharge?
A. NiO(OH) + H2O + 2e- → Ni(OH)2+ OH-
B. Cd + 2OH- → Cd(OH)2 + 2e-
C. Ni(OH)2 + OH- → NiO(OH) + H2O + e-
D. Cd(OH)2 + 2e- → Cd + 2OH-
Solution

5. The following reaction takes place in the experimental cell during discharge.
![]()
A diagram of the experimental cell is shown below.

a. State the energy transformation that occurs in the experimental cell during discharge. 1 mark
Solution
b. Which reactant is the oxidising agent in the experimental cell during discharge? Use oxidation numbers to justify your answer. 2 marks
Solution
c. i. Write the half-equation for the reaction that occurs in the
C14H8O8S2/C14H10O8S2 half-cell during recharge. 1 mark
Solution
ii. State the polarity of the C14H8O8S2/C14H10O8S2 half-cell electrode during recharge. 1 mark
Solution
iii. Explain how the polarity of the electrodes is established during recharge to allow the recharge to occur. 2 marks
Solution
